For example, the atomic weight of beryllium was thought to be approximately 14.5, which would place the element as a homologue of aluminium, whereas Mendeleev argued that his system required the atomic weight to be close to 9. Not only did it predict the existence of new elements, which were subsequently discovered, but it also revised the atomic weight of some of the elements. Second and even more importantly, Mendeleev’s system had a remarkable predictive power. There were basically two major reasons why chemists responded so positively to Mendeleev’s innovation: First, it arranged all the known elements in one coherent system and, thus, established for the first time an organizing principle valid for all the elements and their properties. Advantages of Mendeleev’s Periodic Tableīy the mid-1880s, the periodic system was recognized to be an indispensable key to understand the elements and their combinations into chemical compounds. Predictions, Successes, and Problems 3.1. The periodic system thus has two fathers, but Mendeleev came first and his system was more influential than Meyer’s.įigure 2. It was from this basis that Mendeleev constructed his system of the elements, and it was from the same basis that the German chemist Julius Lothar Meyer independently suggested a roughly similar scheme later in 1869. A pure element could thus be defined as a substance with a particular atomic weight.īy the late 1850s, when Dalton’s ideas had become widely accepted, determinations of atomic weights were an important part of chemistry. Moreover, Dalton introduced the crucial idea of atomic weight, thereby associating an element with a measurable quantity. While this definition is independent of whether matter consists of atoms or not, in 1808 John Dalton took the important step to link together elements and atoms.Īccording to Dalton, an atom is the smallest unit of an element, meaning that there are as many different atoms as there are different elements. Lavoisier’s definition of an element was empirical in so far that he limited elements to substances that could not be decomposed to still simpler substances. The modern concept of an element goes back to the late eighteenth century and owes much to the French chemist Antoine-Laurent Lavoisier, the great reformer of chemistry. Of course, the periodic system presupposes knowledge of what constitutes a chemical element. Dmitri Ivanovich Mendeleev (1834 – 1907) and his periodic table.
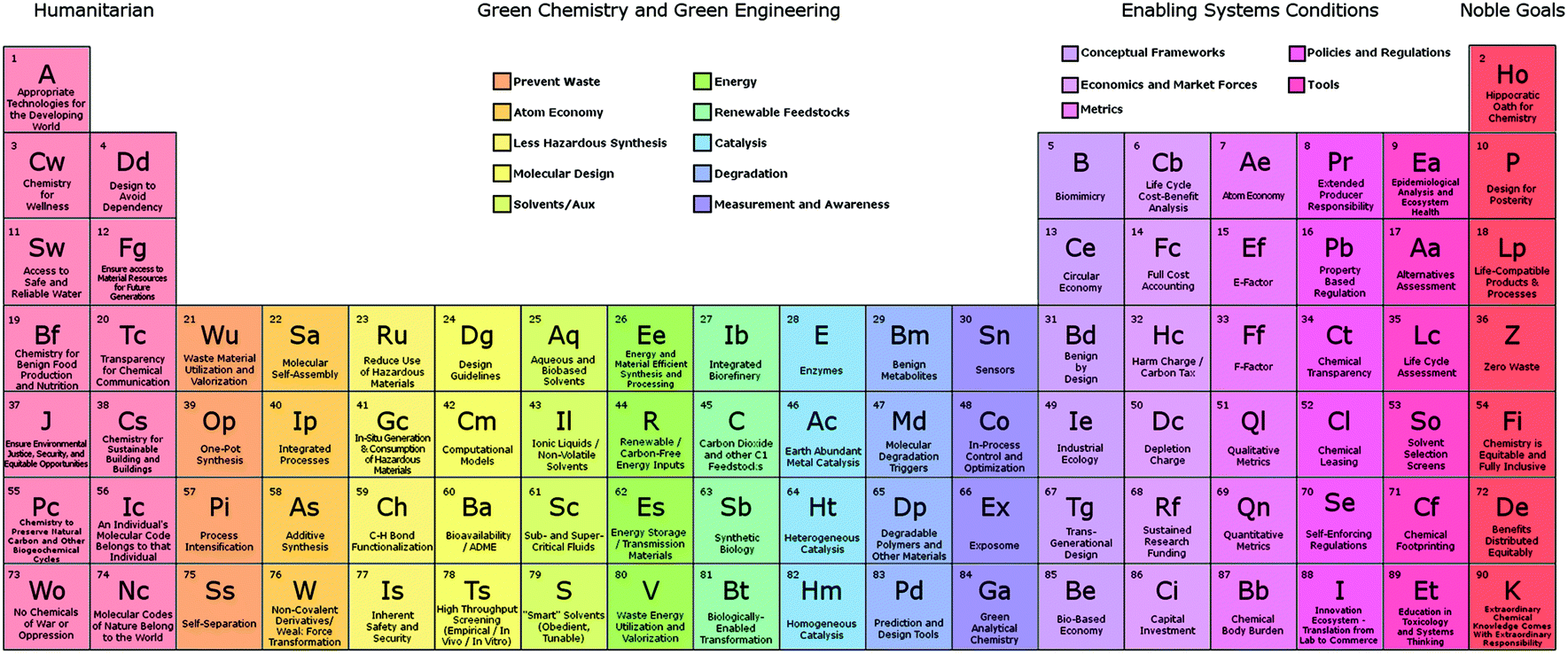
From this point of view, it is as much about physics as it is about chemistry.įigure 1. As it turned out in the 1920s, the periodic system is a macroscopic representation of the internal structure of the atoms. Until the end of the nineteenth century, the periodic system belonged exclusively to the science of chemistry, but when it came to explaining why the system works so well, it were physicists who took the lead. In short, it proved immensely fertile.Īlthough the present versions of the periodic table contain 118 elements compared to the 63 in Mendeleev’s original table, the systems used today descend directly from the one proposed in 1869. Mendeleev’s system or classification of the chemical elements provided inorganic chemistry with a solid foundation which brought order to the confusing amount of experimental data at the same time, it predicted the existence of several hitherto unknown elements. Macroscopic Representation of the Atom’s Internal Structure
I shall use the two terms periodic system or table synonymously.ġ. Mendeleev presented his first version of what subsequently became known as the periodic system or table to the newly established Russian Chemical Society. 150 years ago Dmitri Ivanovich Mendeleev formulated the periodic system, a fundamental law of chemistry which eventually was explained in terms of quantum physics.
